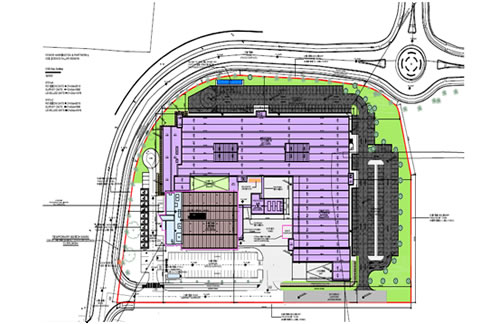
SCOPE: Process Design & CQV
This project involves the fit-out of an existing building shell with a sterile fill-finish suite, inspection, OSD, QC chemistry and microbiology labs, cold store and warehouse.
Equipment included a sterile vial filling line, lyophiliser, autoclave, parts washer, formulation, blender, granulator, mill, press, coater, clean utilities generations skids.
Our scope of services included the following:
- Process flow diagrams
- Process and clean utility design
- Process equipment URSs and technical specifications
- HAZOPs
- pFMEAs
- Tendering of process equipment
- Design support
- CQV of all GMP systems, initially to end of PQ
Please contact linda.nugent@tandempm.ie for futher information.